Rutherford's Theory

Ernest Rutherford (1871-1937) was a physicist from New Zealand who also had great contribution to the development of the atomic model. Many believe that he had the greatest contribution because he discovered the nucleus, protons, neutrons and also received the Nobel Prize in 1908.
Rutherford did an experiment where he directed a stream of alpha particles (positive charge) through a gold foil. He predicted slight direction changes and deflections but would've never thought of what he saw next. Some alpha particles deflected tremendously while others went straight through.
Why did the foil deflect some + particles? --> Atoms must have something positive in them --> But it has to be small because the foil let some + particles stream through --> atoms must have a big empty space with a positive charged center called the nucleus!
Rutherford also assumed that Hydrogen (least massive element) would just have one proton and one electron because it corresponded with its mass. With this theory, Helium (second lightest element) should weigh twice as much as hydrogen. However, it weighed four times the amount of hydrogen, which led to his discovery of neutrons.
Rutherford stated that:
Rutherford did an experiment where he directed a stream of alpha particles (positive charge) through a gold foil. He predicted slight direction changes and deflections but would've never thought of what he saw next. Some alpha particles deflected tremendously while others went straight through.
Why did the foil deflect some + particles? --> Atoms must have something positive in them --> But it has to be small because the foil let some + particles stream through --> atoms must have a big empty space with a positive charged center called the nucleus!
Rutherford also assumed that Hydrogen (least massive element) would just have one proton and one electron because it corresponded with its mass. With this theory, Helium (second lightest element) should weigh twice as much as hydrogen. However, it weighed four times the amount of hydrogen, which led to his discovery of neutrons.
Rutherford stated that:
- Atoms were mainly made up of empty space with a small positively charged center called the nucleus
- The nucleus was made up of protons (positive charge) and neutrons (neutral charge)
- Electrons were located outside of the atom, revolving around it.
Beehive Model
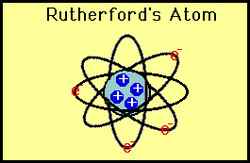
Rutherford's model of an atom included a positively charged center with negative charges orbiting around it. This model is seen very often today, and it's commonly referred to as the "beehive model".
©KarlieTkachankoAnnaZhang
