J.J Thomson's Theory

J.J Thompson (1856-1940) was an English physicist who used the evidence from a conducted experiment to draw conclusions of the atomic model. He was the first person to discover the electron and said that it had a negative charge in 1894. His use of cathode rays emphasized his result since the cathode rays were attracted to the positive plate rather than the negative plate. Therefore, the cathode rays must contain a negative charge.
Thompson stated that:
Thompson stated that:
- Electrons have a negative charge and has less than 1/2000th the mass of a single hydrogen atom.
- Atoms have a net charge of zero
- Atoms are a positive sphere that has a bunch of negative charges (electrons) scattered on it.
Raisin Bun Model
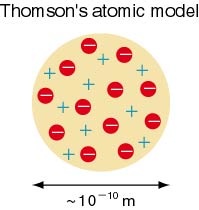
Thomson's viewed the atom as a positively charged sphere with negative charges embedded to balance the charge. This was often called the "plum-pudding" or the "raisin bun" theory because the electrons were scattered like raisins in a raisin bun.
©KarlieTkachankoAnnaZhang
