Bohr's Theory

Even though Rutherford's general idea about the atom model was correct, there was still a question to be answered. If the electrons orbited around the nucleus, then why did the electrons not collapse towards it since opposites attract?
Bohr came up with an explanation.
Niels Bohr (1885-1962) was a Danish physicist who discovered "electron shells" and "energy levels".
Bohr stated that:
Bohr came up with an explanation.
Niels Bohr (1885-1962) was a Danish physicist who discovered "electron shells" and "energy levels".
Bohr stated that:
- Electrons don't just travel randomly, they are on a fixed energy level and orbit in electron shells
- Electrons can only move from one allowed energy to another. They can't just sit in the empty space of two energy levels
- Each energy level can only hold a max number of electrons
Planets orbiting around the sun
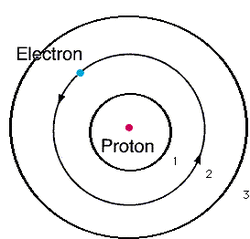
Bohr developed this model with electrons orbiting around the nucleus in electron shells. People tend to make the analogy of this model being like planets orbiting around the sun.
©KarlieTkachankoAnnaZhang
